Photodynamic Therapy for Cancer
Photosensitizers for Photodynamic Therapy
Molecules such as phthalocyanine and their derivatives have strong absorption at around 630 nm wavelength, the region of high permeability through biological tissues. Therefore, they are widely used as photosensitizers for singlet oxygen generation in photodynamic therapy (PDT: a method of cancer treatment in which singlet oxygen is produced by laser irradiation, then, this active singlet oxygen will attack and kill cancer cells). By increasing the light absorption coefficient of sensitizers, the singlet oxygen yield can be improved. However, the strong light absorption of photosensitizers in the tissues on the surface is suggested for the problem that light cannot reach to deep cancer tissues. Therefore, while the photosensitizers are desirable to stably generate singlet oxygen until the completion of treatment, there is a dilemma that they are hoped to decompose after the treatment, so that light can reach to deeper tissues.
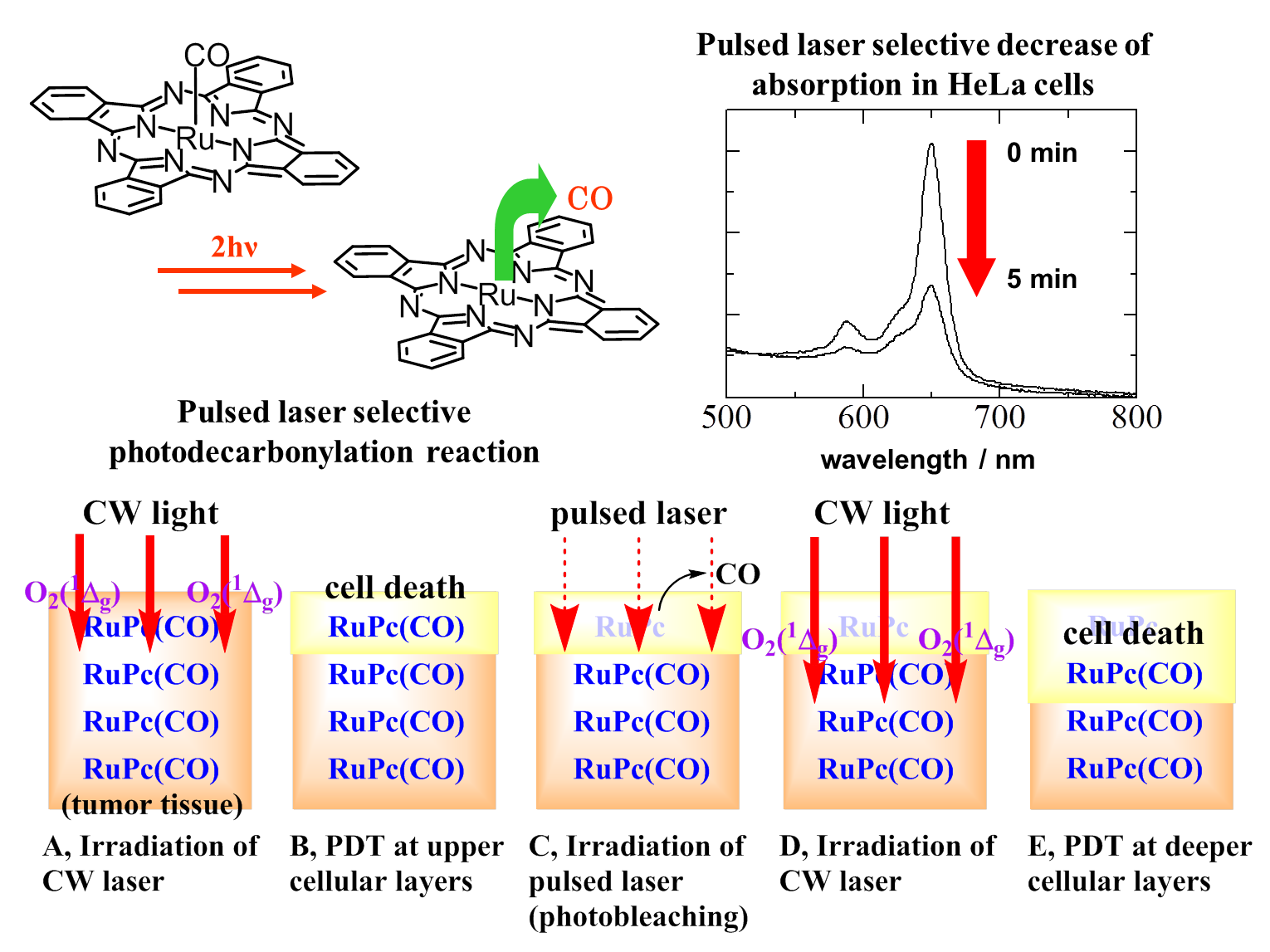
Herein, we proposed ruthenium phthalocyanine (RuPc(CO)(Py)) and naphthalocyanine (RuNc(CO)(Py)) complexes as photosensitizers that satisfied the two above requirements. When a nanosecond-pulsed laser is used, the photodecarbonylation of our Ru complexes efficiently proceeds via stepwise two-photon excitation, while the reaction yields are negligibly small when a continuous-wave (CW) laser is employed. The pulsed laser selective photodecarbonylation decreases the Q-band absorbance, which satisfies what the photodynamic therapy (PDT) requires of the photobleaching. For RuPc(CO)(Py), the photochemical reactions including both the photodecarbonylation and just photobleaching occur in HeLa cells in vitro.
On the basis of these results, we propose a novel concept for achieving a greater depth of necrosis in PDT as follows:
(1) PDT of upper cellular layers using CW laser irradiation;
(2) efficient photobleaching in upper cellular layers using pulsed dye-laser irradiation, which results in an increase in the therapeutic depth of red light;
(3) PDT directed toward deeper tumor tissues using CW laser irradiation.
In addition, these Ru complexes are promising as CO release agents to investigative biochemistry.
Related Papers
-
Kei Murata, Yuki Saibe, Mayu Uchida, Mizuki Aono, Ryuji Misawa, Yoshiho Ikeuchi Kazuyuki Ishii
"Two-Photon, Red Light Uncaging of Alkyl Radicals from Organorhodium(III) Phthalocyanine Complexes"
Chemical Communications, 58, 11280-11283 (2022). -
Kazuyuki Ishii
"Efficient Singlet Oxygen Generation without Heavy-Atom Effect"
Seisan-Kenkyu, 60, 160-163 (2008). -
K. Ishii, M. Shiine, Y. Shimizu, S. Hoshino, H. Abe, K. Sogawa, and N. Kobayashi
"Control of Photobleaching in Photodynamic Therapy Using Photodecarbonylation Reaction of Ruthenium Phthalocyanine Complexes via Stepwise Two-Photon Excitation"
J. Phys. Chem. B, 112, 3138 (2008). -
K. Ishii, A. Takayanagi, S. Shimizu, H. Abe, K. Sogawa, and N. Kobayashi
"In vitro Photodynamic Effects of Phthalocyaninatosilicon Covalently Linked to 2,2,6,6-Tetramethyl-1-Piperidinyloxy Radicals on Cancer Cells"
Free Radical Biol. Med., 38, 920 (2005). -
K. Ishii, H. Itoya, H. Miwa, M. Fujitsuka, O. Ito, and N. Kobayashi
"Relationship between Symmetry of Porphyrinic π-Conjugated Systems and Singlet Oxygen (1Δg) Yields: Low-Symmetrical Tetraazaporphyrin Derivatives"
J. Phys. Chem. A, 109, 5781 (2005). -
K. Ishii, S. Takeuchi, S. Shimizu, and N. Kobayashi
"A Concept for Controlling Singlet Oxygen (1Δg) Yields by Nitroxide Radicals: Phthalocyaninatosilicon Covalently Linked to Nitroxide Radicals"
J. Am. Chem. Soc., 126, 2082 (2004). -
H. Miwa, K. Ishii, and N. Kobayashi
"Electronic Structures of Zinc and Palladium Tetraazaporphyrin Derivatives Controlled by Fused Benzo-Rings"
Chem. Eur. J., 10, 4422 (2004). -
K. Ishii, S. Hoshino, N. Kobayashi
"Photodecarbonylation of Ruthenium Carbonyl Octaethylporphyrin via Stepwise Two-Photon Absorption of Visible Light"
Inorg. Chem., 43, 7969 (2004).
