Electrochemical reduction of CO2

Papers
K. Murata, H. Tanaka and K. Ishii
“Electrochemical Reduction of CO2 by a Gas Diffusion Electrode Composed of fac-Re(diimine)(CO)3Cl and Carbon Nanotubes”
J. Phys. Chem. C, 123, 12073-12080 (2019).
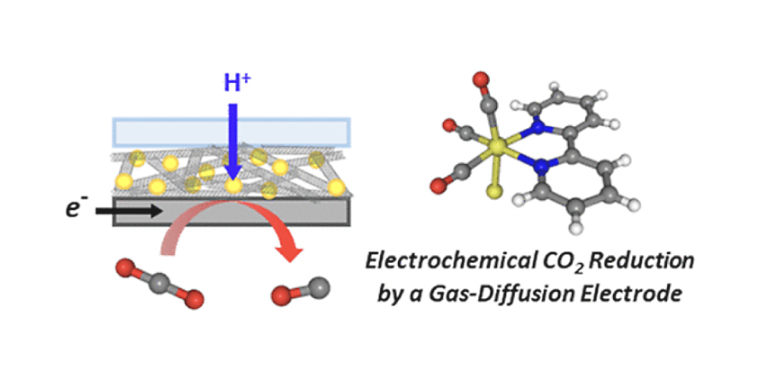
The electrochemical reduction of carbon dioxide (CO2) was investigated using a gas-diffusion electrode containing fac-Re(diimine)(CO)3Cl and carbon nanotubes as the electrocatalyst and its supporting materials, respectively. The catalytic current attributed to CO2 reduction was successfully observed by cyclic voltammetry under a CO2 atmosphere. Electrolysis at a constant potential resulted in the selective conversion of CO2 to CO, which was associated with 2-electron reduction coupled with 2-proton transfers. Screenings of the preparation conditions of the gas-diffusion electrode demonstrated that the catalytic performance strongly depended on the catalyst/carrier ratio, amount of loaded electrode components, and applied electrode potentials. These results indicated that the electrical conductivity and the diffusions of protons and CO2 in the catalyst layer were crucial factors for the reaction efficiency. This study will provide basic strategies for the rational design of gas-diffusion electrode-based electrocatalytic systems for reductions of gas substrates.
Related Papers
- N. T. Hong Trang and K. Ishii
“Photoelectrochemical oxygen reduction reactions using phthalocyanine-based thin films on an ITO electrode”
J. Phys. Chem. C, 122, 3539 (2017).
